Science at Bangor & in the News
|
This week we wrapped up the last of the chemistry units in Science 9. We will start exploring physics when I get back on Tuesday and we'll spend the rest of the year learning about energy, movement, and forces.
We finished up our empirical formula lab in chemistry. Over winter break, I received a donation that covered the cost of 6 Chromebooks, and we used them to share data and discuss the results of our experiment. I'm very excited to be able to continue to use the Chromebooks to help us organize, analyze, and discuss data from our experiments. The end of unit assessment will happen early next week, and then we will begin to learn about the internal structure of atoms. I hope everyone enjoyed their snow days & has a relaxing long weekend!
0 Comments
Even though we only had a 3 day week, both Chemistry & Science 9 worked on some major concepts. Science 9 spent the week working on balancing chemical reactions. This was generally a panic-inducing process at first, but as we kept working on problems and used blocks to represent the atoms in a reaction life got much better. This was the last topic in our interacting particles unit, and we will have the end of unit assessment on Wednesday of this week (2/24).
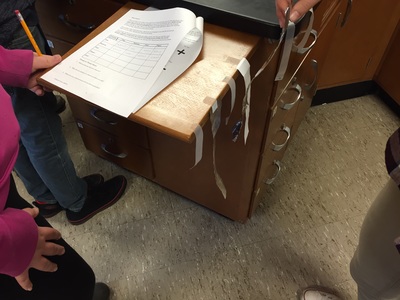
In chemistry we learned about different types of chemical formulas. We spent Tuesday in lab making a salt called zinc chloride and will figure out what the empirical formula for the compound from our lab data. I received a donation over winter break that allowed me to purchase 6 Chromebooks. We are using the Chromebooks to share everyone's data as we collect it and will analyze the data to see if we all come up with the same formulas and discuss how we got our results & why they might be different between groups. This week in chemistry, we entered the world of moles. Moles in chemistry are a unit of particles, kind of like a dozen, not the rodent who ruins your yard. After spending a couple of days working on converting between mass, moles, & number of particles, we used the end of the week to put those skills to the test. Everyone got paired up and had to figure out how much mass was in a certain number of particles (or moles) of salt, water, and aluminum foil. The pairs also got samples of copper & had to figure out how many particles were in the sample. I think it was good for everyone to see how much (or how little) of an actual sample were in the very large numbers we were dealing with. In Science 9, we spent the weak learning about chemical reactions. We used cubes to represent different compounds and then modeled what happened when those compounds either changed from liquid to gas or had something happen that caused the atoms to be rearranged. We used these changes as a starting point to compare and contrast physical changes and chemical changes. We ended the week talking about why we need to write balanced equations and how to balance equations. This is our last topic in the unit & after this unit we will move on to an introduction to physics. You may have seen something about gravitational waves in the news this week. Gravitational waves are distortions in time and space that were first proposed to exist by Albert Einstein in 1915 when he published his theory on general relativity. Many scientists working together for many years were finally able to detect these waves in September of 2015. The distortion the laser detected is 1000 times smaller than the width of a proton! You can read about how the waves were detected here. To learn more about what gravitational waves are, go here. We've really gotten back into the swing of things this week. In Science 9, we spent the first part of the week putting together & giving presentations on various elements of the periodic table. Highlights included a titanium trivia poster, a bilingual magnesium poster, an arsenic pop-up book, several poems, and excellent bromine models. Some of these will be on display at Bangor Creates, so be on the lookout! On Thursday & Friday we began to add to our particle model. During the Sticky Tape Lab, we observed attractions and repulsions between charged objects. To explain what happened, we used Thomson's model of the atom: a positively charged sphere with small, negatively charged particles (electrons) stuck inside the sphere. We can think of this model like a blueberry muffin. The positively charged atom is like the cake part of the muffin and the electrons are like the blueberries. We will move forward with atomic models and begin to investigate chemical reactions. In chemistry, we learned about relative mass (how many times heavier one element is than another) and how compounds will always have the same mass ratio. Different compounds can be made of the same elements, just in a different ratio. One example of this is water (2 hydrogens + 1 oxygen) and hydrogen peroxide (2 hydrogens + 2 oxygens). We also started our next unit on counting particles by getting the mass of sets of hardware. Particles are very tiny, so we need a different way to count them. Since particles of an element are always the same, we can use mass to count the number of particles. We can also compare the relative mass of particles by comparing how many times larger one element is than another if the same number of particles are present. We have a couple of labs coming up that will help us with the idea of counting particles by mass.
|
Ms. Lyons
Science is amazing, check it out! Archives
May 2016
Categories |






















 RSS Feed
RSS Feed
