Science at Bangor & in the News
 This week we spent the week balancing reactions. We practiced balancing the reactions using a few different methods: using cubes to represent the atoms in each compound & making the products from the cubes or trying to get equal numbers of each cube on both sides of the reactions, drawing out particle representations of the atoms and balancing the numbers of particles, and counting up the number of each atom without drawing or using cubes. Each student picked their favorite method & worked from there. At the end of the week we added an additional challenge: starting with a written out reaction, having to write the chemical formulas, and finally balancing the reaction. This puts together skills from our last unit (naming chemical compounds) with balancing reactions. Science 9 is just about finished with our unit on forces. This week we discussed Newton's 2nd law (Force = mass * acceleration), reviewed the rest of the unit, and took the end of unit assessment. On Friday, we watched videos on the physics and biology of car crashes in order to prepare for our project for the unit: An egg drop. We will be building egg protection containers that will hopefully protect raw eggs from being cracked after being dropped off of the band tower.
0 Comments
After a 2 week hiatus, chemistry is back in session! This week we conducted a 3 day long experiment. We added iron nails to copper chloride salt dissolved in water. The next day, the nails and solution looked very different than when we started! We scraped off the solid produced into a beaker and let the solid and the nails dry overnight. The third day we took the mass of everything & tried to make sense of what had happened. We decided that the iron from the nails had switched place with the dissolved copper ions and wrote out the following reaction: Fe + CuCl2 --> Cu + FeCl2. After the lab, we discussed what happens in chemical reactions--atoms get rearranged. We also started exploring how reactions work in terms of conserving mass and atoms. Science 9 spent the week learning about Newton's 3rd law: "For every reaction there is an equal and opposite reaction." We did a series of demonstrations in class with pairs of students holding & pulling a spring scale (Thanks to Bloomingdale's Mrs. Wenger for loaning us the spring scales!). We could see that no matter who was pulling or who was holding, the scales always read the same amount. We used this as a basis for drawing out how the forces balanced out in force diagrams. Next week we will be wrapping up the material for this unit & then we will be on to a fun project. Stay tuned for the project announcement!
Unfortunately, chemistry did not really meet at all this week because of state testing. We will start back at it tomorrow with an experiment to kick off our unit on representing chemical change.
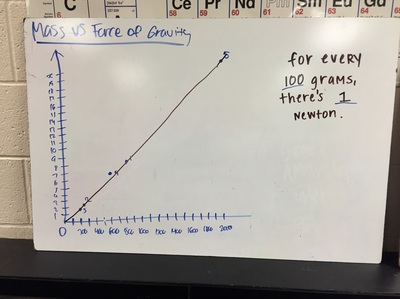
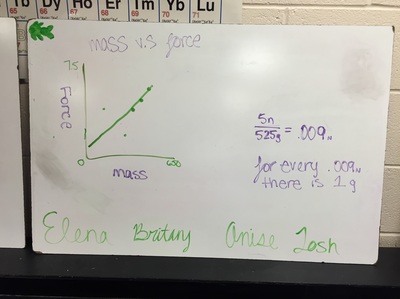
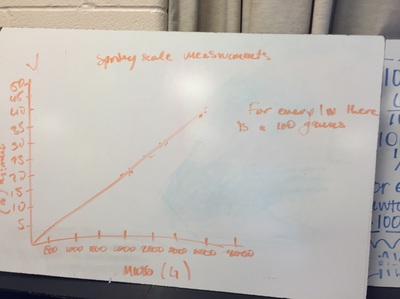
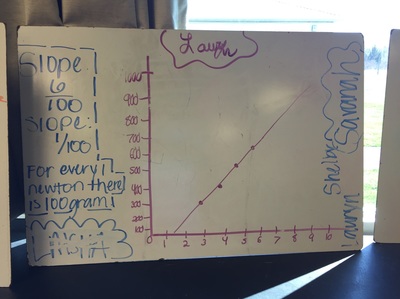
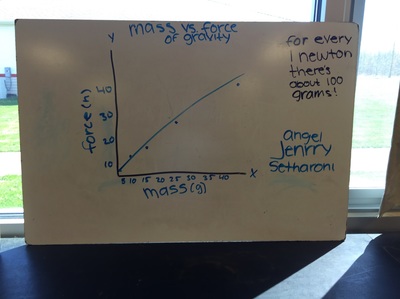
Science 9 did meet last week. We continued our unit on forces by working on how to represent forces is different situations. The most difficult part was probably remembering which of our 7 (!) forces was being used. We will continue to work on the vocabulary and identifying forces as the unit progresses. At the end of the week we started focusing on the force of gravity. We used spring scales (Thank you, Ms. Wenger!) to measure the mass and weight (force of gravity) on various objects that were around the classroom. When we analyzed our data, most groups had very similar constants for the force of gravity. During our discussion we agreed that this made sense since we are all on the same planet and gravity is the same no matter where we are on Earth. Our data showed that the force of gravity on Earth is about 0.01 Newtons for each gram, or 10 Newtons for each kilogram. The actual force of gravity is 9.81 Newtons for each kilogram...I was very impressed with how close we were able to get with simple spring scales. |
Ms. Lyons
Science is amazing, check it out! Archives
May 2016
Categories |


















 RSS Feed
RSS Feed
