Science at Bangor & in the News
|
I can't believe the year is almost over. We have 7 days of regular school and 3 days of finals left. Chemistry is officially senior-less as of Wednesday, so the end of the week felt a bit empty without them. We will be wrapping up our last topic this week and then we will review for the final exam. Science 9 will be finishing up their last unit assessment on Tuesday. The next few days after will be a review to prepare for the final. Both classes will be taking their free-response portion of their finals in class on Tuesday, June 7 and Wednesday, June 8. The time during final exams will be to take the multiple choice portion. The final exam schedule is below.
Thursday, June 9-1st hr (2nd & 3rd hr review), 4th hr (5th & 6th hr review) Friday, June 10-2nd hr& 3rd hr (1/2 day for students) Monday, June 13-5th hr & 6th hr (1/2 day for students)
0 Comments
Chemistry is inching toward our Diet Coke & Mentos geyser day & just wrapped up our chemical reactions unit. We are now starting to learn about how much stuff is involved in chemical reactions & how to use the balanced reaction to figure out how much reactant we need or how much product we can make. In order to introduce this idea, we made s'mores. We needed 2 graham crackers, 1 marshmallow, and 3 pieces of Hershey's chocolate to make 1 s'more. We knew how many of each ingredient we had, so we could figure out how many whole s'mores we could make. It was a tasty way to begin this unit. We have moved on to start solving problems with chemical reactions. In Science 9 we are continuing to explore motion. This week we moved from a new way to represent an object's motion visually to distinguishing objects with constant velocity from those with constant acceleration. We can all picture a car maintaining its velocity (speed) or a car speeding up, but it is a little more difficult to determine what is happening just by looking at a graph. As we continue to practice analyzing graphs, more light bulbs are going on & we are starting to connect what is happening on the graph to what an object would actually be doing.
We wrapped up our chemistry unit on interacting particles by talking about energy in reactions. We used energy bar charts (affectionately known as LoL charts since that's what the charts look like) to show what happens to energy in a chemical reaction. We were able to show transferring energy between the thermal energy account (how fast the particles move) and the chemical energy account (how the atoms in a molecule are arranged). We also showed how energy can come into a reaction system from outside or be released into the surroundings from the reaction. This is a complicated process to learn and I'm very proud of how the students kept sticking with it & asking questions until they understood what was happening & why reactions happen the way they do.
In Science 9, we continued our study of motion using tumble buggies. This week we started the buggies from different points & sent them in the opposite direction of one another. We used this to start talking about how it is more useful to show an object's position at certain times instead of just how far an object has gone from its starting point. We will continue to learn about motion for the next couple of weeks. We performed 2 different experiments this week in chemistry. Our first experiment looked at 5 different types of chemical reactions. We were able to perform one set of experiments in lab, I demonstrated another, and we used a YouTube video from a friend to view the other 3 types of experiments. Two of our previous labs were also examples of one of the types of reactions. The next day, we talked about when we actually use some of these reactions in our daily life. After some thinking, we came up with good examples of the different reaction types. Our second lab let us observe & feel the difference between endothermic reactions and exothermic reactions. Endothermic reactions need energy put into them to make them happen. We observed a reaction that used the thermal energy from the room temperature water to happen, so we could see (with a thermometer) and feel the reaction tube get colder as the water transferred its energy to the compound. An exothermic reaction is just the opposite; it releases energy into the surroundings. We saw & felt the reaction tube get warmer as the reaction happened. Most people are familiar with exothermic reactions: the reason we feel warm next to a fire is that the combustion reaction that causes the fire gives off it's extra energy & raises the temperature around the reaction. On Thursday we wrapped up our forces unit in Science 9 by testing our egg protection containers. Mrs. Doe kindly let us drop the containers from the marching band viewing tower. It was a great day to spend some time outside & drop some eggs. All of the eggs survived the fall, although we did lose 1 egg on extraction. The containers all had some common themes, but there were also differences among them. It was great watching the students use their knowledge of forces and collisions to creatively come up with a solution. The last unit we are covering is looking at motion. We started the unit on Friday by measuring how far a toy buggy went in a certain amount of time or how long it took the buggy to travel a certain distance. The weather actually cooperated and we were able to test the buggies outside. We haven't looked at the data yet, but we will refresh our graphing skills starting on Monday when we start analyzing the data.
 This week we spent the week balancing reactions. We practiced balancing the reactions using a few different methods: using cubes to represent the atoms in each compound & making the products from the cubes or trying to get equal numbers of each cube on both sides of the reactions, drawing out particle representations of the atoms and balancing the numbers of particles, and counting up the number of each atom without drawing or using cubes. Each student picked their favorite method & worked from there. At the end of the week we added an additional challenge: starting with a written out reaction, having to write the chemical formulas, and finally balancing the reaction. This puts together skills from our last unit (naming chemical compounds) with balancing reactions. Science 9 is just about finished with our unit on forces. This week we discussed Newton's 2nd law (Force = mass * acceleration), reviewed the rest of the unit, and took the end of unit assessment. On Friday, we watched videos on the physics and biology of car crashes in order to prepare for our project for the unit: An egg drop. We will be building egg protection containers that will hopefully protect raw eggs from being cracked after being dropped off of the band tower. After a 2 week hiatus, chemistry is back in session! This week we conducted a 3 day long experiment. We added iron nails to copper chloride salt dissolved in water. The next day, the nails and solution looked very different than when we started! We scraped off the solid produced into a beaker and let the solid and the nails dry overnight. The third day we took the mass of everything & tried to make sense of what had happened. We decided that the iron from the nails had switched place with the dissolved copper ions and wrote out the following reaction: Fe + CuCl2 --> Cu + FeCl2. After the lab, we discussed what happens in chemical reactions--atoms get rearranged. We also started exploring how reactions work in terms of conserving mass and atoms. Science 9 spent the week learning about Newton's 3rd law: "For every reaction there is an equal and opposite reaction." We did a series of demonstrations in class with pairs of students holding & pulling a spring scale (Thanks to Bloomingdale's Mrs. Wenger for loaning us the spring scales!). We could see that no matter who was pulling or who was holding, the scales always read the same amount. We used this as a basis for drawing out how the forces balanced out in force diagrams. Next week we will be wrapping up the material for this unit & then we will be on to a fun project. Stay tuned for the project announcement!
Unfortunately, chemistry did not really meet at all this week because of state testing. We will start back at it tomorrow with an experiment to kick off our unit on representing chemical change.
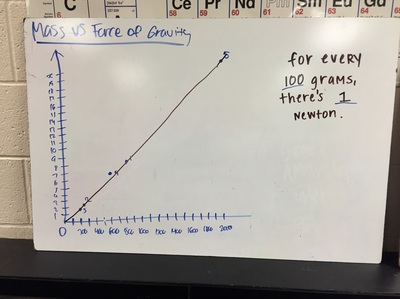
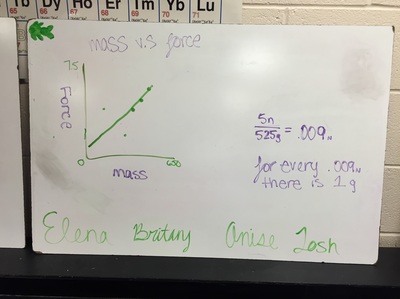
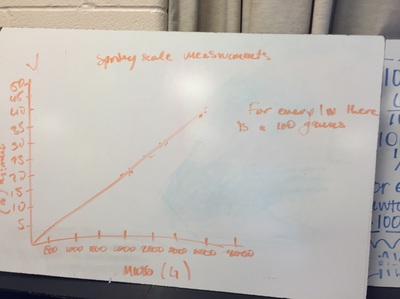
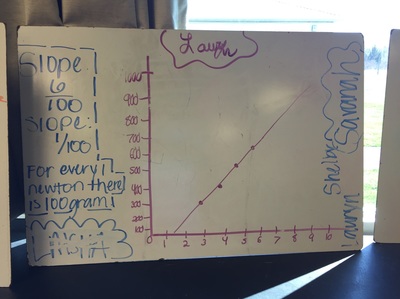
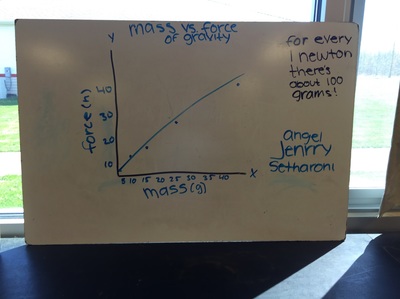
Science 9 did meet last week. We continued our unit on forces by working on how to represent forces is different situations. The most difficult part was probably remembering which of our 7 (!) forces was being used. We will continue to work on the vocabulary and identifying forces as the unit progresses. At the end of the week we started focusing on the force of gravity. We used spring scales (Thank you, Ms. Wenger!) to measure the mass and weight (force of gravity) on various objects that were around the classroom. When we analyzed our data, most groups had very similar constants for the force of gravity. During our discussion we agreed that this made sense since we are all on the same planet and gravity is the same no matter where we are on Earth. Our data showed that the force of gravity on Earth is about 0.01 Newtons for each gram, or 10 Newtons for each kilogram. The actual force of gravity is 9.81 Newtons for each kilogram...I was very impressed with how close we were able to get with simple spring scales. We are a half-day away from Spring Break as I type this. Chemistry just wrapped up our unit on the internal structure of atoms. We learned about electrons, charges, and how different types of compounds have different structures. We wrapped up the unit by learning how to name different types of compounds. After the juniors get through testing, we will start a unit that explores what chemical reactions are and different types of chemical reactions. We will observe different types of reactions and then use our particle model to explain what happened. I'm really looking forward to this unit because we will get to perform some fun labs and try to make sense of our observations.
In Science 9 we wrapped up a unit on energy last week and started a unit on forces. We ended the week by beginning to represent forces in a diagram. Once we get all of the new vocabulary down, I think we will begin to have a lot of fun with this unit. We will wrap this unit up with an egg drop. The students will be given some design constraints (volume, mass, no parachutes/wings/etc) and will design and build a container that will keep the egg in tact after being dropped from either the crow's nest or the gym roof. On our half-day last week, 5th hour Science 9 began working with their new groups. In order to start working as a team, the groups were challenged to save Fred. Fred (the gummy worm) was out on Lake Michigan in his boat (a cup) when it capsized. Fred was able to get on top of the boat, but his lifesaver (a gummy Lifesaver) was stuck under the boat. The teams needed to use their rescue tools (paperclips) to get Fred off the boat, right the boat, & then get Fred into his lifesaver & back into his boat. The teams had to write directions on how they made the rescue. They switched instructions with another team and needed to perform the rescue again while strictly following the other team's instructions. This was a very fun exercise for everyone, and also a very important one in science. Scientists communicate their discoveries in writing. Part of new ideas being accepted in the scientific community is repeatability. If other scientists are not able to repeat the original work, that original work gets called into question. Scientists need to communicate what they discovered and exactly how they discovered it. I think having to follow another group's instructions for saving Fred emphasized how important written communication in science is. It has certainly been a while! I apologize for the long absence. Life got a little hectic and then I managed to catch some sort of virus. Since the last update, Science 9 has been studying energy. We started out by thinking about how energy is involved in different situations that the students likely experienced before (dropping a ball, using a pull-back or wind-up toy, rubbing hands together). After learning about the different ways that energy can be stored or transferred, the students are putting their new knowledge to the test by building Rube Goldberg machines. For a great example, watch this video for Goldie Blox. Chemistry wrapped up a unit about counting particles & then began investigating internal structure. We used interactions involving static electricity to deduce that there are charged particles inside atoms. We are now starting to learn about how electrons influence how compounds form and how the structure of compounds affects their properties (strength, melting point, boiling point, etc.). In other exciting news, my GoFundMe campaign to purchase new equipment is fully funded! I now have 6 new ChromeBooks and 6 sets of Vernier instruments that will interface with the ChromeBooks. These tools will allow us to perform more experiments, collect better data, and share data with each other as we collect it. I am so grateful to everyone who donated and shared the campaign and am looking forward to helping the students experience how the process of science actually works.
This week we wrapped up the last of the chemistry units in Science 9. We will start exploring physics when I get back on Tuesday and we'll spend the rest of the year learning about energy, movement, and forces.
We finished up our empirical formula lab in chemistry. Over winter break, I received a donation that covered the cost of 6 Chromebooks, and we used them to share data and discuss the results of our experiment. I'm very excited to be able to continue to use the Chromebooks to help us organize, analyze, and discuss data from our experiments. The end of unit assessment will happen early next week, and then we will begin to learn about the internal structure of atoms. I hope everyone enjoyed their snow days & has a relaxing long weekend! |
Ms. Lyons
Science is amazing, check it out! Archives
May 2016
Categories |





































































 RSS Feed
RSS Feed
