Science at Bangor & in the News
|
Even though we only had a 3 day week, both Chemistry & Science 9 worked on some major concepts. Science 9 spent the week working on balancing chemical reactions. This was generally a panic-inducing process at first, but as we kept working on problems and used blocks to represent the atoms in a reaction life got much better. This was the last topic in our interacting particles unit, and we will have the end of unit assessment on Wednesday of this week (2/24).
In chemistry we learned about different types of chemical formulas. We spent Tuesday in lab making a salt called zinc chloride and will figure out what the empirical formula for the compound from our lab data. I received a donation over winter break that allowed me to purchase 6 Chromebooks. We are using the Chromebooks to share everyone's data as we collect it and will analyze the data to see if we all come up with the same formulas and discuss how we got our results & why they might be different between groups.
0 Comments
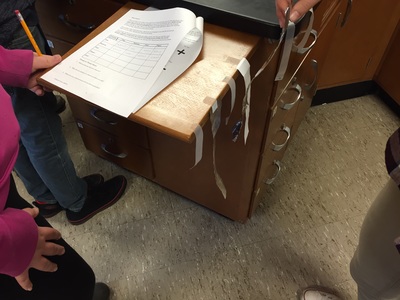
This week in chemistry, we entered the world of moles. Moles in chemistry are a unit of particles, kind of like a dozen, not the rodent who ruins your yard. After spending a couple of days working on converting between mass, moles, & number of particles, we used the end of the week to put those skills to the test. Everyone got paired up and had to figure out how much mass was in a certain number of particles (or moles) of salt, water, and aluminum foil. The pairs also got samples of copper & had to figure out how many particles were in the sample. I think it was good for everyone to see how much (or how little) of an actual sample were in the very large numbers we were dealing with. In Science 9, we spent the weak learning about chemical reactions. We used cubes to represent different compounds and then modeled what happened when those compounds either changed from liquid to gas or had something happen that caused the atoms to be rearranged. We used these changes as a starting point to compare and contrast physical changes and chemical changes. We ended the week talking about why we need to write balanced equations and how to balance equations. This is our last topic in the unit & after this unit we will move on to an introduction to physics. You may have seen something about gravitational waves in the news this week. Gravitational waves are distortions in time and space that were first proposed to exist by Albert Einstein in 1915 when he published his theory on general relativity. Many scientists working together for many years were finally able to detect these waves in September of 2015. The distortion the laser detected is 1000 times smaller than the width of a proton! You can read about how the waves were detected here. To learn more about what gravitational waves are, go here. We've really gotten back into the swing of things this week. In Science 9, we spent the first part of the week putting together & giving presentations on various elements of the periodic table. Highlights included a titanium trivia poster, a bilingual magnesium poster, an arsenic pop-up book, several poems, and excellent bromine models. Some of these will be on display at Bangor Creates, so be on the lookout! On Thursday & Friday we began to add to our particle model. During the Sticky Tape Lab, we observed attractions and repulsions between charged objects. To explain what happened, we used Thomson's model of the atom: a positively charged sphere with small, negatively charged particles (electrons) stuck inside the sphere. We can think of this model like a blueberry muffin. The positively charged atom is like the cake part of the muffin and the electrons are like the blueberries. We will move forward with atomic models and begin to investigate chemical reactions. In chemistry, we learned about relative mass (how many times heavier one element is than another) and how compounds will always have the same mass ratio. Different compounds can be made of the same elements, just in a different ratio. One example of this is water (2 hydrogens + 1 oxygen) and hydrogen peroxide (2 hydrogens + 2 oxygens). We also started our next unit on counting particles by getting the mass of sets of hardware. Particles are very tiny, so we need a different way to count them. Since particles of an element are always the same, we can use mass to count the number of particles. We can also compare the relative mass of particles by comparing how many times larger one element is than another if the same number of particles are present. We have a couple of labs coming up that will help us with the idea of counting particles by mass.
This week kicked off the 2nd half of the school year. We spent time reviewing last semester in both Chemistry & Science 9. Science 9 spent the 2nd half of the week researching an element of their choice. The poems, comic strips, models, & other products of the element research project will be displayed for Bangor Creates! We will use what we learn about the elements to start learning about atoms in more detail and how atoms interact.
In chemistry, we are wrapping up the unit we didn't quite finish before finals. We are working on trying to figure out a chemical formula based on the ratio of the masses of the elements in the compound. From here we will start talking about how chemists use mass to count the number of particles. This semester flew by! It doesn't seem like all that long ago that school started & I was just trying to learn everyone's names. I hope that everyone had a positive science experience so far and I'm looking forward to what we will experience & learn next semester.
After reviewing what we learned before break, we spent the week in chemistry describing matter and learning how units of matter come together. We talked about how mixtures, compounds, and elements are different at the level we can see and at the particle level. One of the characteristics that we can observe is solubility. Some substances will dissolve in other substances, like sugar dissolving in water. However, there are some substances, like ethanol, that sugar won't dissolve in. The photos below show a sugar cube that I placed in ethanol during first hour on Tuesday (1/5). The last photo was taken today (1/8) after school. So if we have a sample of water & one of ethanol, but don't know which is which, we could throw a sugar cube in each & see which one sugar dissolves into. If sugar dissolves, the sample is water. If the sugar cube stays whole, the sample is ethanol. We also learned that compounds have distinct characteristics from the substances used to form the compound, but in mixtures the components keep their characteristics and the overall characteristics of the mixture are an average of the components. The beaker on the left (W) has water with a fully dissolved sugar cube (I promise there was a sugar cube dropped in it!), the beaker in the middle (E) is the same beaker of ethanol shown above, and the beaker on the right (50) has a 50/50 mixture of ethanol and water. Some of the sugar dissolved, but there is still a pile of sugar on the bottom of the beaker (it's on the right side of the beaker). The big news in chemistry from over winter break is that chemists just synthesized the elements to fill out the last (7th) row of the periodic table. There were 4 elements missing that were just approved by the International Union of Pure and Applied Chemistry (IUPAC). The elements were made by scientists in Japan, the US, and Russia. The scientists that made the elements get to give the elements their permanent names. Within the next 6 months, the elements will be named & a new official periodic table will be issued. For more information, see the article from Chemical & Engineering News.
We began the last unit before the final exam in Science 9. Students were able to observe particles moving. After I lit a candle in one corner of the room, the scientists nearest the candle could smell the candle first, and gradually the students further and further away could smell the candle too. If particles didn't move, the smell particles would never have gotten to the other side of the room. Dropping food coloring in warm and cold water at the same time showed us that particles at warmer temperatures move faster than particles at cooler temperatures (pictures below). The food coloring quickly spread in the flask with warm water and took much longer to spread in the flask with cold water. We spent the last part of the week talking about pressure. Sucking the air particles out of a trash bag that a student volunteer was sitting in caused the bag to give the volunteer a hug (picture below). The air particles in the room were bombarding the bag, but without air particles in the bag to push back, the bag was pushed against the volunteer. It was a big week in the classroom & in chemistry! We've got next week and part of the week after before we take final exams. I hope everyone is starting to look over their notes from early in the semester, eating healthy, & getting plenty of sleep in preparation for exams. Have a great weekend, Vikings (and everyone else)! Both chemistry & science 9 wrapped up the units they were working on. When we get back from break, we will review what we've learned so far this year & start working on new material before final exams. Exams are January 20-22. The students will be in school all day on Wednesday, January 20 and will only report for the morning on Thursday, January 21, and Friday, January 22. Hopefully we will not loose a week to snow & cold this year like we have the past few years.
After we finished our units, both classes spent the past two days being crafty & playing science games. I enjoyed seeing the students' creativity as we made ornaments using a Borax solution. When we get back from break we will discuss how and why the crystals formed in the Borax solution. Here are some highlights below. In Science 9 we wrapped up the unit on particles & matter. We spent the week on density: the amount of stuff (matter) in a certain amount of space (volume). This week we began using the relationship between density, mass, & volume to solve problems using algebra. This idea was a challenging one, so we will practice problem solving some more next week before the end of unit assessment. We wrapped up the week with a lab practical. Each group chose a sample of material and needed to identify what the material was made out of. In order to be successful, the groups had to accurately measure mass and volume so that they could calculate the density.
Chemistry finished the unit on energy and states of matter and took the end of unit assessment on Friday. Before the assessment, we learned about how much energy gets absorbed or released in systems. To do this, we took what we were doing with the LOL charts & started to put numbers to the energy flow. This was very challenging and I am very proud of how hard the students worked to understand what we were doing. I'm sorry for the late update. Last weekend I performed with the Kalamazoo Bach Festival Choir in our Christmas concert, so I didn't get a chance to update the blog.
In chemistry we spent the week talking about how energy affects how particles move & how they are arranged. We illustrated the flow within energy accounts using energy bar charts, also known as LOL diagrams. The students did a great job learning to work with this new tool and I think it helped some people get a better understanding of how energy going into or coming out of the different energy accounts changes what is happening to the particles. At the end of the week we learned about how we can figure out the quantity of energy going into or out of a system. We are continuing to work on energy calculations this week. We did two different labs last week in Science 9. The first lab looked at how mass does or doesn't change depending on the experiment. This lab allowed us to discover the Law of Conservation of Mass: mass in a system doesn't change unless we add or subtract stuff (matter). Our second lab looked at how mass & volume are related to each other. We spent today looking at our data and talking about density. We will be wrapping up the unit this week and will have the Unit 4 assessment on Wednesday next week. Below are some pictures from both the mass and change lab and the mass and volume lab. After some technical difficulties last week, we were able to do our experiment in chemistry on Monday. We chilled water to 0 degrees Celsius, then heated it up to boiling, and recorded the temperature over the course of the experiment. Nearly every group hit a point in the experiment where no matter how long the water sat on the hot plate, the temperature would not increase. We learned that there are a couple of reasons the temperature stopped increasing: 1. We would have needed a special chamber to measure the temperature of the steam coming off of the water. 2. The energy that the hot plate was providing wasn't going into the thermal account anymore (used to increase temperature), it was going into the phase account instead (used to increase the space between particles). We also talked about how the meaning of heat scientists use (heat = energy) often gets confused with how we use heat outside of the classroom (heat = warm temperature), so from now on we will not be using the word heat so we can be clear when we are talking about our observations. In Science 9, we started talking about mass, the amount of stuff in an object, and particles. Each group of students took a pair of objects that were the same size, but one of the objects was lighter than the other. We decided that the heavier object had more stuff, and probably had a higher number of particles, than the lighter object. We will continue to explore more about mass and particles this unit. Our first experiment examining what happens to mass when we change things will be on Tuesday. |
Ms. Lyons
Science is amazing, check it out! Archives
May 2016
Categories |

























































 RSS Feed
RSS Feed
