Science at Bangor & in the News
|
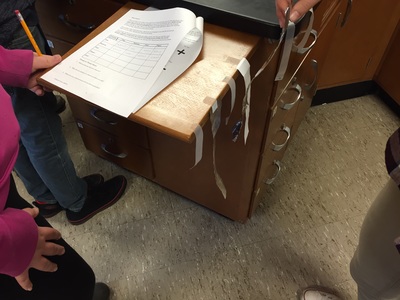
We've really gotten back into the swing of things this week. In Science 9, we spent the first part of the week putting together & giving presentations on various elements of the periodic table. Highlights included a titanium trivia poster, a bilingual magnesium poster, an arsenic pop-up book, several poems, and excellent bromine models. Some of these will be on display at Bangor Creates, so be on the lookout! On Thursday & Friday we began to add to our particle model. During the Sticky Tape Lab, we observed attractions and repulsions between charged objects. To explain what happened, we used Thomson's model of the atom: a positively charged sphere with small, negatively charged particles (electrons) stuck inside the sphere. We can think of this model like a blueberry muffin. The positively charged atom is like the cake part of the muffin and the electrons are like the blueberries. We will move forward with atomic models and begin to investigate chemical reactions. In chemistry, we learned about relative mass (how many times heavier one element is than another) and how compounds will always have the same mass ratio. Different compounds can be made of the same elements, just in a different ratio. One example of this is water (2 hydrogens + 1 oxygen) and hydrogen peroxide (2 hydrogens + 2 oxygens). We also started our next unit on counting particles by getting the mass of sets of hardware. Particles are very tiny, so we need a different way to count them. Since particles of an element are always the same, we can use mass to count the number of particles. We can also compare the relative mass of particles by comparing how many times larger one element is than another if the same number of particles are present. We have a couple of labs coming up that will help us with the idea of counting particles by mass.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Ms. Lyons
Science is amazing, check it out! Archives
May 2016
Categories |







 RSS Feed
RSS Feed
